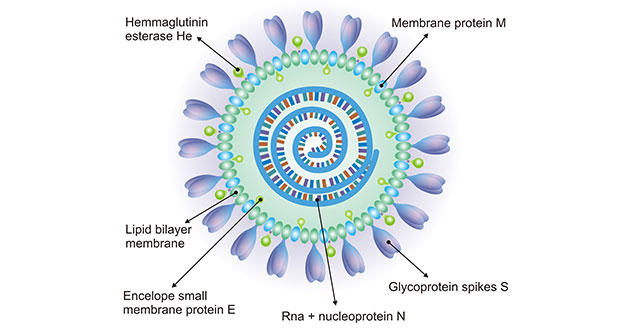
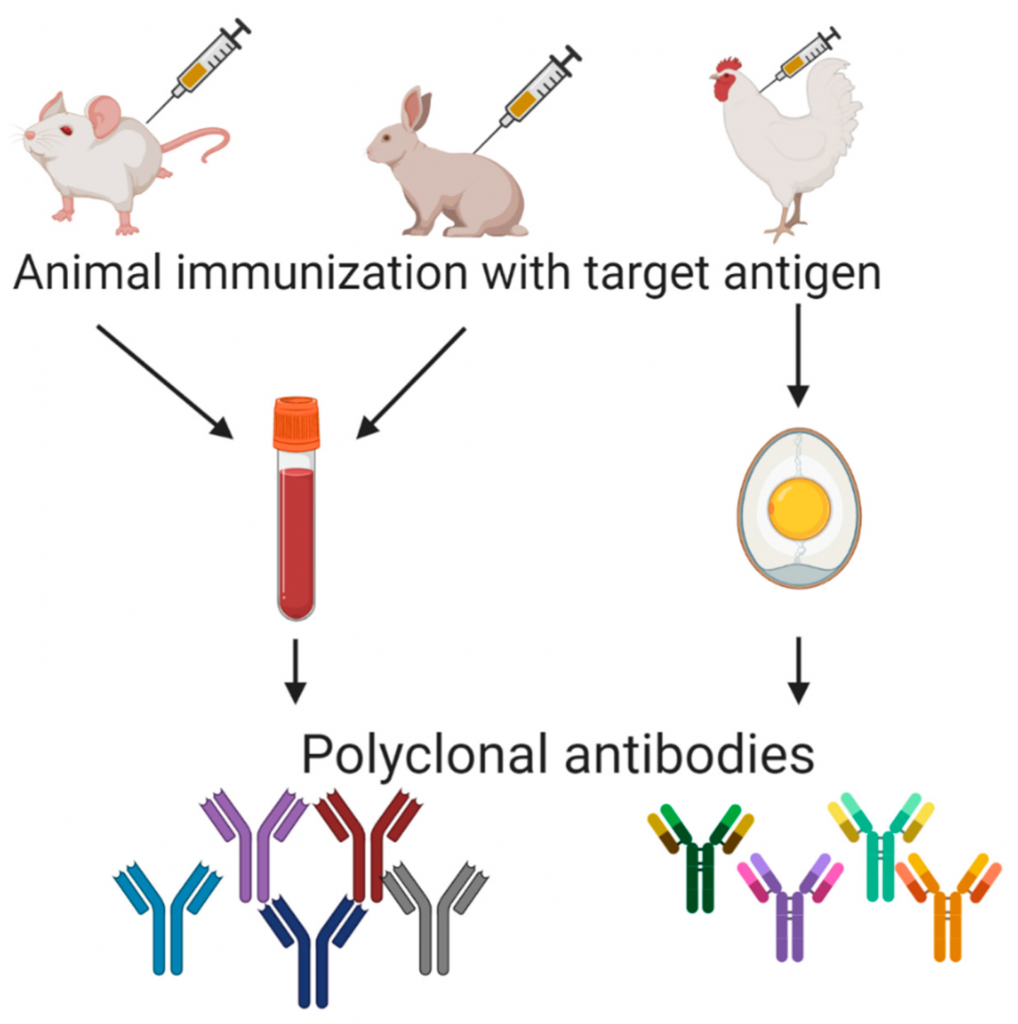
Cusabio CoronaVirus Proteins
Abstract Since the COVID-19 pandemic, more than two million worldwide have died from the virus. To address one of the biggest challenges we face, numerous researchers are making unprecedented efforts to combat COVID-19. To support COVID-19 research, CUSABIO has developed a large number of reagents related to SARS-CoV-2, including antibody, protein, and ELISA kits. Here […]
Cusabio CoronaVirus Proteins Read More »